 The dipole moment is an important property to consider in various chemical reactions and physical interactions involving polar molecules.Hint: The size of the P atom is greater than the N atom, down the group basicity of the atom decreases when the size of the atom increases and the electron density decreases. Dipole moments occur when there is a separation of charge. 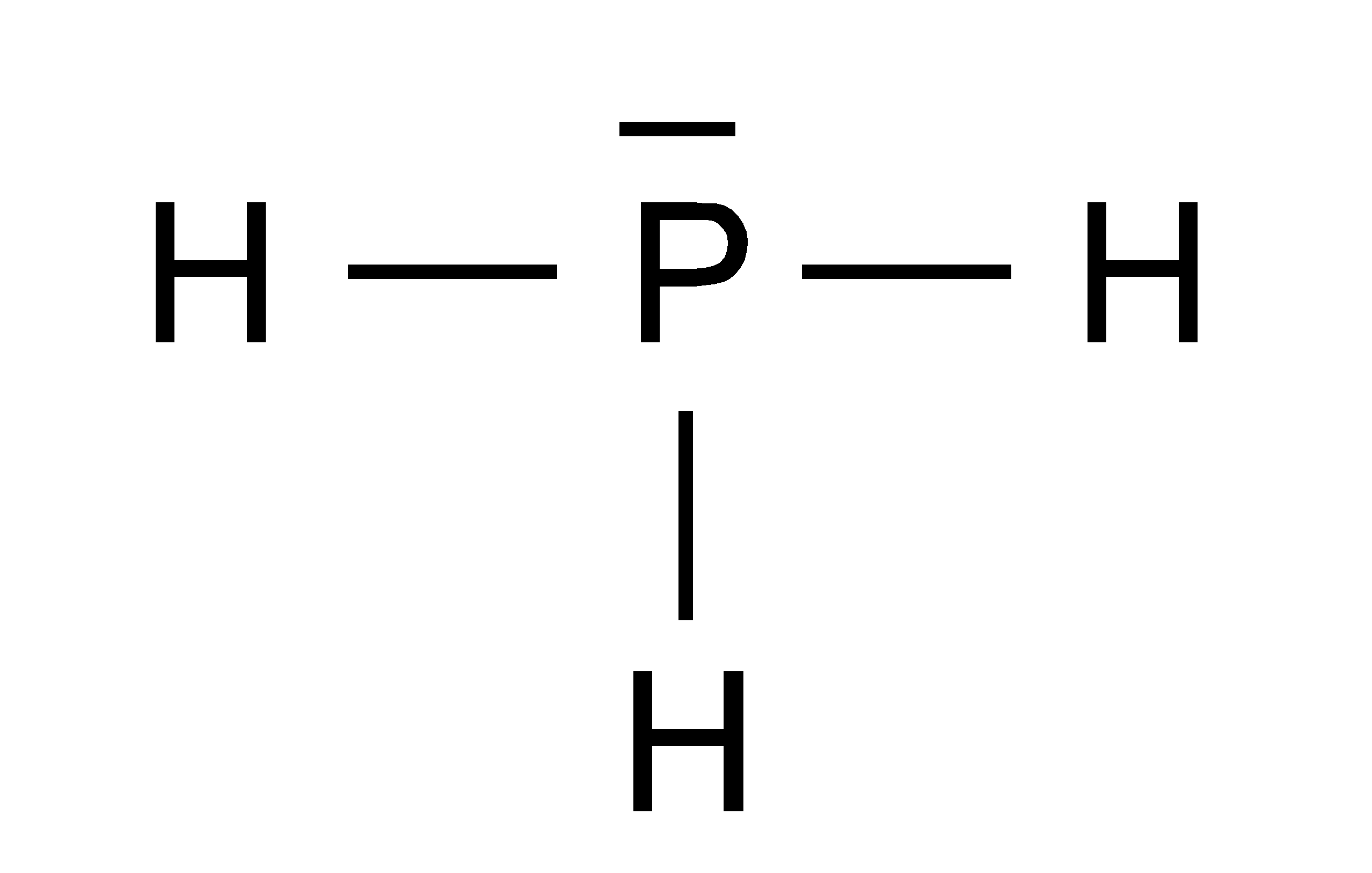
It forms dipole-dipole because it is a polar molecule. oxygen is more electronegative than hydrogen). Why are dipole dipole forces permanent The permanent dipole in water is caused by oxygen’s tendency to draw electrons to itself (i.e. It occurs whenever positive and negative charges are separated. In conclusion, the dipole moment of water (H2O) is greater than that of ammonia (NH3) due to the higher electronegativity difference and shorter distance between charges in water. A molecule with a permanent dipole moment is called a polar molecule. The bond dipole moment is a measurement of the polarity of a chemical bond within a molecule that is based on the electric dipole moment concept. O COS has a dipole moment because it is asymmetric and the dipole moments do not completely cancel. Choose one: O COS has no dipole moment because the molecule is linear and dipole moments cancel in linear molecules. (b) Which molecule do you expect to have a larger dipole moment Sections 11.2 and 11.5. Does COS have a dipole moment Choose the answer that best explains the reason for your choice. 
In ammonia, the nitrogen atom is less electronegative than oxygen, resulting in a larger distance between the charges and a smaller dipole moment compared to water. Determine if phosphorus trihydride, PH3, is polar or nonpolar. This ratio, together with the computed shielding constants, was used to determine a new value of the nuclear magnetic dipole moment of (31)P: (P) 1.1309246(. This results in a shorter distance between the positive and negative charges, further enhancing the dipole moment. In water, the oxygen atom is more electronegative, causing the electron density to be pulled closer to the oxygen atom. The dipole moment is also affected by the distance between the charges. This greater electronegativity difference in water leads to a greater charge separation, resulting in a larger dipole moment compared to ammonia. In water (H2O), the electronegativity difference between oxygen and hydrogen atoms is higher than the electronegativity difference between nitrogen and hydrogen atoms in ammonia (NH3). The dipole moment of a molecule is influenced by two factors: the magnitude of the charge separation and the distance between the charges. This separation of charges creates a dipole moment in the molecule. The nitrogen atom is more electronegative than the hydrogen atoms, leading to a partial negative charge on the nitrogen atom and partial positive charges on the hydrogen atoms. Similarly, ammonia is also a polar molecule due to its pyramidal molecular geometry and the electronegativity difference between nitrogen and hydrogen atoms. The oxygen atom is more electronegative than the hydrogen atoms, resulting in a partial negative charge on the oxygen atom and partial positive charges on the hydrogen atoms. C) The electrons in a polar bond are found nearer to the more electronegative element. 
B) Dipole moments result from the unequal distribution of electrons in a molecule. Water is a polar molecule due to its bent molecular geometry and the electronegativity difference between oxygen and hydrogen atoms. 3.4 (12 reviews) Which of the following statements is incorrect A) Ionic bonding results from the transfer of electrons from one atom to another. In this case, we are comparing the dipole moments of water (H2O) and ammonia (NH3). A dipole moment occurs when there is a separation of positive and negative charges in a molecule. Accurate values of the dipole moments ( v2 1 0.5740 0.0002 D) and of the transition frequencies have been obtained. It is defined as the product of the charge (Q) and the distance (d) between the charges in a molecule. Nine transitions belonging to the v4 and v4 bands of PH 3 have been brought in resonance with CO 2 laser lines, using an intracavity Stark cell. The dipole moment is a measure of the polarity of a molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
